Protein Purification Optimisation at Ab Biotechnology
At Ab Biotechnology we have extensive experience in purification of recombinant and native source proteins, and both monoclonal and polyclonal antibodies.

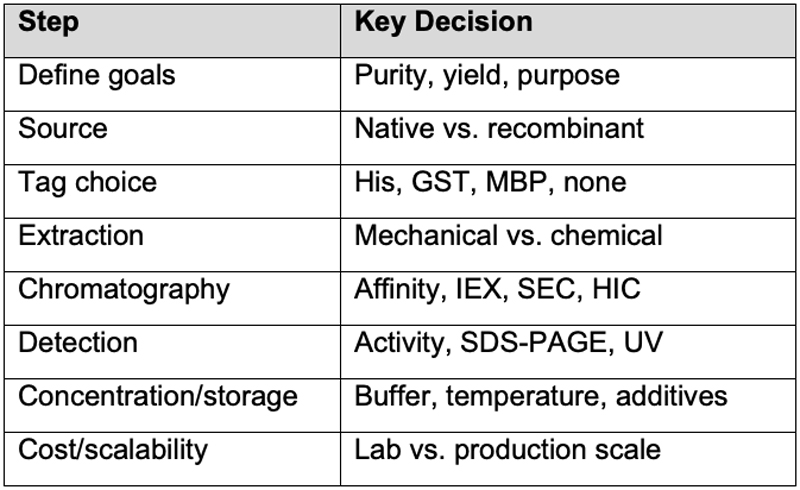
What are our key steps and decisions for protein production?
When planning protein purification, several key decisions determine the success and efficiency of the process and ultimately how pure your final protein preparation will be.
1. Define the Goal
Before doing anything:
- Purpose of purification: analytical (small scale), structural (e.g. crystallography), or functional (activity assay, therapeutics)?
- Required purity: crude extract, partially purified, or >95% pure?
- Yield vs. purity trade-off: how much purity can you afford to lose for higher yield?
- Are there existing methods for production of the protein: small-scale development work, published methods?

2. Source of the Protein
Decide where your protein comes from:
- Natural source (e.g. tissue, bacteria, plants): more complex mixtures, lower yield.
- Recombinant expression (e.g. E. coli, yeast, mammalian cells): allows tagging and easier purification.
Tagged vs. untagged: affinity tags (His-tag, GST, MBP) simplify purification but might affect activity.
3. Solubility and Stability
- Soluble or insoluble protein? Soluble proteins are easier; insoluble proteins require solubilisation and refolding. A refolding screen can identify the optimal conditions.
- Buffer conditions: pH, salt concentration, cofactors, reducing agents.
- Protease inhibitors: prevent degradation during purification.
4. Lysis and Extraction Method
- Mechanical disruption: sonication, French press, homogenization.
- Chemical/enzymatic: lysozyme, detergents.
- Temperature control: keep everything cold to prevent denaturation and proteolysis.
5. Purification Strategy (Chromatography Steps)
Choose separation methods based on protein properties:


Typically: Capture → Intermediate → Polishing (e.g. affinity → ion exchange → size exclusion)

6. Detection and Monitoring
How will you detect your protein throughout the process?
- Absorbance at 280 nm
- Enzymatic activity assay
- SDS-PAGE / Western blot
- Mass spectrometry
7. Concentration and Storage
- Concentration method: ultrafiltration, lyophilization, Tangential Flow Filtration.
- Storage conditions: buffer, pH, cryoprotectants (e.g. glycerol), temperature (4°C, -20°C, -80°C).
- Stability tests: freeze-thaw cycles, oxidation sensitivity, heat, light.
8. Scalability and Cost
- Can the method be scaled up or automated?
- How expensive are the columns/reagents?
- Is the tag removable (via protease cleavage) if needed?
- Is GMP manufacture required?
Summary Table
Please get in contact if you would like to see how we can support your projects.
enquiries@abbiotechnology.com

